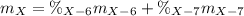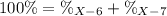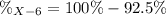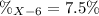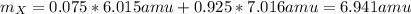Chemistry, 22.10.2020 20:01 BreBreDoeCCx
Element X has two natural isotopes: X-6 (6.015 amu) and X-7 (7.016 amu). Calculate the atomic mass of element X given the abundance of X-7 is 92.5%

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:10
The rock in a lead ore deposit contains 89 % pbs by mass. how many kilograms of the rock must be processed to obtain 1.5 kg of pb?
Answers: 1

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2


You know the right answer?
Element X has two natural isotopes: X-6 (6.015 amu) and X-7 (7.016 amu). Calculate the atomic mass o...
Questions



Computers and Technology, 18.02.2020 18:25







Mathematics, 18.02.2020 18:26

Computers and Technology, 18.02.2020 18:26

Mathematics, 18.02.2020 18:26

Mathematics, 18.02.2020 18:26



Chemistry, 18.02.2020 18:26