
Chemistry, 23.10.2020 03:01 braines2003
The rate constant for the reaction below was determined to be 3.241×10-5 s–1 at 800 K. The activation energy of the reaction is 255 kJ/mol. What would be the value of the rate constant at 9.50×102 K?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Perform the following mathematical operations and report the answer to the appropriate number of significant figures 5.87998 + 3.100
Answers: 2

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2
You know the right answer?
The rate constant for the reaction below was determined to be 3.241×10-5 s–1 at 800 K. The activatio...
Questions



Computers and Technology, 01.03.2021 21:50



Advanced Placement (AP), 01.03.2021 21:50

Mathematics, 01.03.2021 21:50

Social Studies, 01.03.2021 21:50

SAT, 01.03.2021 21:50


Social Studies, 01.03.2021 21:50


Mathematics, 01.03.2021 21:50





Chemistry, 01.03.2021 21:50


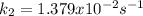
![k_2=k_1exp[-\frac{Ea}{R}(\frac{1}{T_2}-\frac{1}{T_1} )]](/tpl/images/0834/1791/1a893.png)
![k_2=3.241x10^{-5}s^{-1}exp[-\frac{255000}{8.314}(\frac{1}{950}-\frac{1}{800} )]\\\\k_2=1.379x10^{-2}s^{-1}](/tpl/images/0834/1791/1944c.png)


