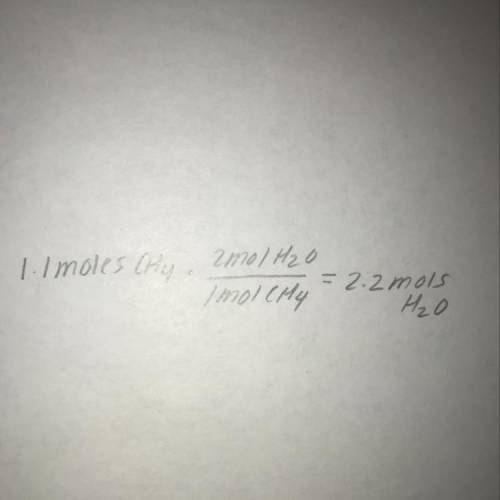QUESTION 1
Consider the following reaction: CH4 + 202 --> 2H2O + CO2
How many moles of wat...


Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1
You know the right answer?
Questions


English, 02.04.2020 18:16



English, 02.04.2020 18:16


Mathematics, 02.04.2020 18:16

Mathematics, 02.04.2020 18:16

Mathematics, 02.04.2020 18:16

Mathematics, 02.04.2020 18:16

Mathematics, 02.04.2020 18:16

Mathematics, 02.04.2020 18:17

Mathematics, 02.04.2020 18:17


Mathematics, 02.04.2020 18:17



Mathematics, 02.04.2020 18:17

Geography, 02.04.2020 18:17