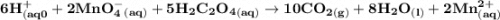Chemistry, 26.10.2020 16:40 Hannahdavy5434
A student dissolved a 0.139g sample of oxalic acid, H2C2O4, in water in an Erlenmeyer flask. Then the student titrated the H2C2O4 solution in the flask with a solution of KMnO4, which has a dark purple color. The balanced chemical equation for the reaction that occurred during the titration is shown above. (a) Identify the species that was reduced in the titration reaction. Justify your answer in terms of oxidation numbers.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1
You know the right answer?
A student dissolved a 0.139g sample of oxalic acid, H2C2O4, in water in an Erlenmeyer flask. Then th...
Questions

Mathematics, 23.11.2021 16:10


Mathematics, 23.11.2021 16:10



Mathematics, 23.11.2021 16:10

English, 23.11.2021 16:10


English, 23.11.2021 16:10

Mathematics, 23.11.2021 16:10

History, 23.11.2021 16:10


Mathematics, 23.11.2021 16:10

English, 23.11.2021 16:10

Geography, 23.11.2021 16:10

Health, 23.11.2021 16:10

Mathematics, 23.11.2021 16:10

Mathematics, 23.11.2021 16:10

Mathematics, 23.11.2021 16:10