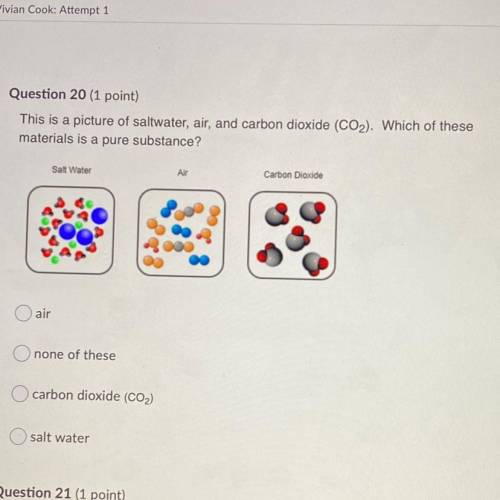
I ALSO NEED HELP WITH THIS ITS MY UNIT TEST PLEASE
...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
Questions

Business, 18.09.2019 15:00


Mathematics, 18.09.2019 15:00

Mathematics, 18.09.2019 15:00


Social Studies, 18.09.2019 15:00


History, 18.09.2019 15:00

Mathematics, 18.09.2019 15:00


History, 18.09.2019 15:00

Mathematics, 18.09.2019 15:00

Mathematics, 18.09.2019 15:00


Mathematics, 18.09.2019 15:00



English, 18.09.2019 15:00

History, 18.09.2019 15:00

Advanced Placement (AP), 18.09.2019 15:00