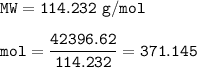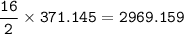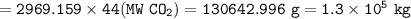Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2


Chemistry, 23.06.2019 04:40
Temperature is defined as a. the equivalent of heat. b. a measure of the average kinetic energy of the individual atoms or molecules composing a substance. c. how hot or cold it is. d. the total kinetic energy of the atoms or molecules composing a substance. e. none of the above is correct.
Answers: 1
You know the right answer?
What mass of CO2 (in kilograms) does the combustion of a 16-gallon tank of gasoline release into the...
Questions

Engineering, 18.11.2020 04:50

Social Studies, 18.11.2020 04:50


Mathematics, 18.11.2020 04:50




Mathematics, 18.11.2020 04:50

Mathematics, 18.11.2020 04:50


Biology, 18.11.2020 04:50




Mathematics, 18.11.2020 04:50

Mathematics, 18.11.2020 04:50


Engineering, 18.11.2020 04:50

Mathematics, 18.11.2020 04:50

Mathematics, 18.11.2020 04:50