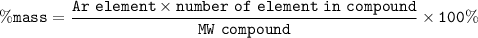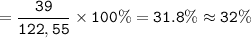Chemistry, 28.10.2020 15:50 andybiersack154
Breanne needed to find the percent composition of the compound KClOz The percent of potassium in the compound yes approximately A) 29% 32% 39% 43% Hydrogen peronde is a harmful by product of normal metabolic activity. In order to prevent damage, b, drogen peroxide mus: be

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the formula of the ionic compound composed of calcium cations and chloride anions
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Breanne needed to find the percent composition of the compound KClOz The percent of potassium in the...
Questions


Mathematics, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10

Biology, 23.12.2020 18:10




Mathematics, 23.12.2020 18:10






English, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10

Biology, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10


Mathematics, 23.12.2020 18:10

English, 23.12.2020 18:10