
Chemistry, 29.10.2020 16:30 saraaaaaaaa20
what is the hydronium ion concentration in a solution formed by combining 750mL of 0.10M NaOH with 250mL of 0.30 M HCl

Answers: 2


Another question on Chemistry

Chemistry, 20.06.2019 18:02
When undergoing chemical reactions where does the reactant combine to the enzyme?
Answers: 1

Chemistry, 21.06.2019 21:30
Put these processes of the water cycle in the correct order, starting at the point where the water is in the lake: 1. water evaporates into the atmosphere 2. rain, snow, or other precipitation falls 3. water collects into larger bodies of water 4. water vapor condenses into liquid water
Answers: 1

Chemistry, 21.06.2019 22:00
What is driving behind plate tectonics (plate movment)? a) gravity only b) inertia c) convection and gravity d) the sun theres no option for science so i picked chemistry. plz
Answers: 2

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1
You know the right answer?
what is the hydronium ion concentration in a solution formed by combining 750mL of 0.10M NaOH with 2...
Questions


Mathematics, 23.12.2020 20:10

Chemistry, 23.12.2020 20:10




Mathematics, 23.12.2020 20:10


Mathematics, 23.12.2020 20:10

Chemistry, 23.12.2020 20:10

Biology, 23.12.2020 20:10

Social Studies, 23.12.2020 20:10


Mathematics, 23.12.2020 20:10

Biology, 23.12.2020 20:10


Mathematics, 23.12.2020 20:10

Mathematics, 23.12.2020 20:10

English, 23.12.2020 20:10

Biology, 23.12.2020 20:10

![[H^+]=1x10^{-7}M](/tpl/images/0851/2686/77707.png)

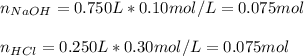
![[H^+]=10^{-pH}=10^{-7}=1x10^{-7}M](/tpl/images/0851/2686/cdb5d.png)


