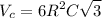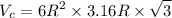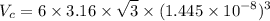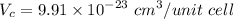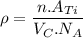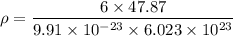Chemistry, 29.10.2020 17:10 estefaniapenalo
Titanium has an HCP unit cell for which the ratio of the lattice parameters c/a is 1.58. If the radius of the Ti atom is 0.1445 nm, calculate the density of Ti and compare it with the literature value of 4.51 g/cm3 . The atomic mass of titanium is 47.87 g/mol.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2


Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3
You know the right answer?
Titanium has an HCP unit cell for which the ratio of the lattice parameters c/a is 1.58. If the radi...
Questions


Mathematics, 22.01.2021 19:50

Computers and Technology, 22.01.2021 19:50


Social Studies, 22.01.2021 19:50













Mathematics, 22.01.2021 19:50