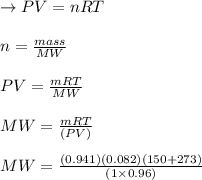A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of , and it weighs . Assuming the gas behaves as an ideal gas under these conditions, calculate the molar mass of the compound. Be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 21:00
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

You know the right answer?
A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of...
Questions




Biology, 24.10.2019 08:43

Computers and Technology, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43




Health, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43



Mathematics, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43


English, 24.10.2019 08:43

Mathematics, 24.10.2019 08:43

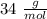 "
"