VERY EASY
PLEASE HELP 10 POINTS
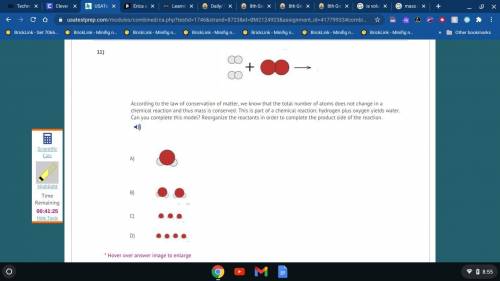
...

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1

Chemistry, 23.06.2019 10:30
Most ionic compouds are crystalline solids at room temperature. true falseionic compounds are electrically neutral. true falseionic compounds generally have low melting points. true falsewhen melted, ionic compounds do not conduct electricity. true falsethe electrostatic attraction between an anion and a cation is an ionic bond. true false
Answers: 1
You know the right answer?
Questions

Business, 15.07.2019 02:20

English, 15.07.2019 02:20

Biology, 15.07.2019 02:20

Social Studies, 15.07.2019 02:20

Biology, 15.07.2019 02:20

History, 15.07.2019 02:20



Biology, 15.07.2019 02:20

Biology, 15.07.2019 02:20



Mathematics, 15.07.2019 02:20

Biology, 15.07.2019 02:20

Social Studies, 15.07.2019 02:20

Social Studies, 15.07.2019 02:20



Biology, 15.07.2019 02:20

Social Studies, 15.07.2019 02:20