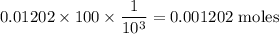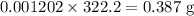Chemistry, 03.11.2020 16:30 elwinelwin9475
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M. Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 1.00

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2
You know the right answer?
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid m...
Questions

English, 04.12.2021 08:00



History, 04.12.2021 08:00


Mathematics, 04.12.2021 08:00

Chemistry, 04.12.2021 08:00

Mathematics, 04.12.2021 08:00

Mathematics, 04.12.2021 08:00

Mathematics, 04.12.2021 08:00

World Languages, 04.12.2021 08:00

History, 04.12.2021 08:00

Chemistry, 04.12.2021 08:00



History, 04.12.2021 08:00

Mathematics, 04.12.2021 08:00



History, 04.12.2021 08:00

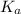 = Acid dissociation constant =
= Acid dissociation constant = 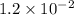
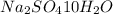 )
)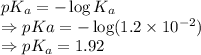
![pH=pK_a+\log\dfrac{[A^-]}{[HA]}\\\Rightarrow pH-pK_a=\log\dfrac{[A^-]}{[HA]}\\\Rightarrow 10^{pH-pK_a}=\dfrac{[A^-]}{[HA]}\\\Rightarrow [A^-]=10^{pH-pK_a}[HA]\\\Rightarrow [A^-]=10^{1-1.92}\times0.1\\\Rightarrow [A^-]=0.01202\ \text{M}](/tpl/images/0863/3238/f5459.png)