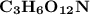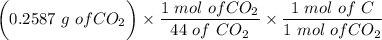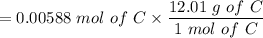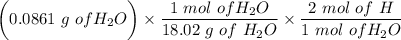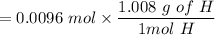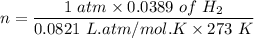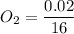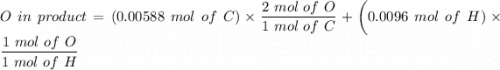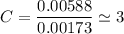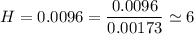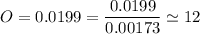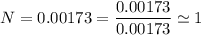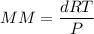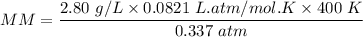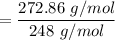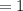An organic compound contains , , , and . Combustion of 0.1023 g of the compound in excess oxygen yielded 0.2587 g and 0.0861 g . A sample of 0.4831 g of the compound was analyzed for nitrogen by the Dumas method. The compound is first reacted by passage over hot : The product gas is then passed through a concentrated solution of to remove the . After passage through the solution, the gas contains and is saturated with water vapor. At STP, 38.9 mL of dry was obtained. In a third experiment, the density of the compound as a gas was found to be 2.86 g/L at 127°C and 256 torr. What are the empirical and molecular formulas of the compound? (Enter the elements in the order: C, H, N, O.)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3


Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
You know the right answer?
An organic compound contains , , , and . Combustion of 0.1023 g of the compound in excess oxygen yie...
Questions

Geography, 30.07.2021 21:50

History, 30.07.2021 21:50


Mathematics, 30.07.2021 21:50


Mathematics, 30.07.2021 21:50

Mathematics, 30.07.2021 21:50

History, 30.07.2021 21:50


Mathematics, 30.07.2021 21:50


Arts, 30.07.2021 21:50

English, 30.07.2021 21:50

Mathematics, 30.07.2021 21:50

Mathematics, 30.07.2021 21:50


Mathematics, 30.07.2021 21:50


Biology, 30.07.2021 21:50

English, 30.07.2021 21:50