
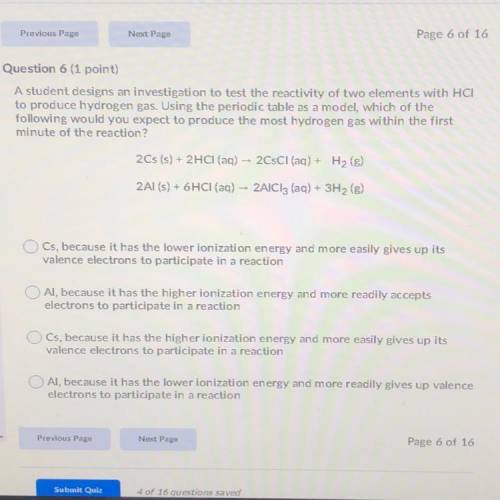
A student designs an investigation to test the reactivity of two elements with HCI
to produce hydrogen gas. Using the periodic table as a model, which of the
following would you expect to produce the most hydrogen gas within the first
minute of the reaction?
2Cs (s) + 2HCl(aq) -- 2CsCl (aq) + H2(8)
2Al(s) + 6HCl(aq) -- AICI, (aq) + 3H2 (8)
Cs, because it has the lower lonization energy and more easily gives up its
valence electrons to participate in a reaction
Al, because it has the higher lonization energy and more readily accepts
electrons to participate in a reaction
Cs, because it has the higher lonization energy and more easily gives up its
valence electrons to participate in a reaction
Al, because it has the lower ionization energy and more readily gives up valence
electrons to participate in a reaction

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 23.06.2019 10:30
Describe the hybridization of each carbon and nitrogen atom in each of the following structures
Answers: 1

Chemistry, 23.06.2019 11:00
Just on number 2 (all parts), and if you do answer explain in detail
Answers: 3
You know the right answer?
A student designs an investigation to test the reactivity of two elements with HCI
to produce hydro...
Questions

Spanish, 07.05.2021 06:00



Mathematics, 07.05.2021 06:00



Medicine, 07.05.2021 06:00




Mathematics, 07.05.2021 06:00

Chemistry, 07.05.2021 06:00

Mathematics, 07.05.2021 06:00

Mathematics, 07.05.2021 06:00

Mathematics, 07.05.2021 06:00


Physics, 07.05.2021 06:00



Mathematics, 07.05.2021 06:00



