
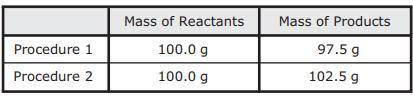
As part of an investigation, students combined substances in a beaker to observe chemical reactions. They performed two procedures. They measured the mass of each substance before and after each reaction. The table shows their observations.
Assuming the students did not make any careless errors, what likely explains these changes in mass?
A. Procedure 1: All the reactants were liquids that evaporated.
Procedure 2: A gas was formed as one product, and it escaped into the air.
B. Procedure 1: One of the reactants was converted to thermal energy.
Procedure 2: All the products were liquids.
C. Procedure 1: The reactants were liquids with different densities.
Procedure 2: The reactants were combined into only one product.
D. Procedure 1: One of the products was a gas that escaped into the air.
Procedure 2: A gas from the air reacted with one of the other reactants.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 23.06.2019 00:50
Which of the following warnings would an agricultural chemist tell a farmer who wants to recycle his or her own ammonia? recycling ammonia is a difficult process that sometimes takes weeks. recycling ammonia requires a degree in biochemistry or a related field. recycling ammonia can be harmful because it is highly flammable and toxic. recycling ammonia costs too much money considering the price of the necessary chemicals.
Answers: 1

Chemistry, 23.06.2019 12:30
The equilibrium constant kc for the reaction 2 nocl(g) → 2 no(g) + cl2(g) is 0.453 at a certain temperature. a mixture of nocl, no, and cl2 with concentrations 1.30, 1.20, and 0.600 m, respectively, was introduced into a container at this temperature. which of the following is true? 1. no apparent reaction takes place. 2. [cl2] = 0.30 m at equilibrium. 3. nocl(g) is produced until equilibrium is reached. 4. [nocl] = [no] = [cl2] at equilibrium. 5. cl2(g) is produced until equilibrium is
Answers: 3

Chemistry, 23.06.2019 12:50
Which of these describes the rate of this chemical reaction? h2 + cl2 → 2 hcl a. an increase in the concentration of hcl and h2 with time b. an increase in the concentration of hcl with time c. an increase in h2 and cl2 with time d. a decrease in hcl and cl2 with time
Answers: 1
You know the right answer?
As part of an investigation, students combined substances in a beaker to observe chemical reactions....
Questions

English, 09.11.2020 23:20


Mathematics, 09.11.2020 23:20



Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20


Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20

Social Studies, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20


Mathematics, 09.11.2020 23:20

Physics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20

Mathematics, 09.11.2020 23:20



