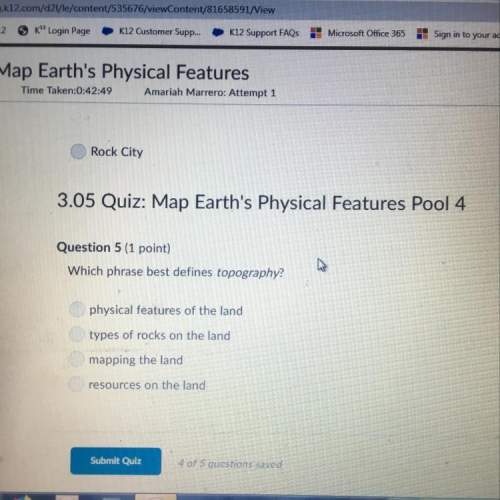
Chemistry, 05.11.2020 18:30 haylieshaven1
Initially, only A and B are present, each at 2.00 M . What is the final concentration of A once equilibrium is reached?
What is the final concentration of D at equilibrium if the initial concentrations are [A] = 1.00M and [B] = 2.00 M?
The reversible chemical reaction A + B ↔ C + D has the following equilibrium constant: Kc = [C][D]/[A][B] = 4.7.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

You know the right answer?
Initially, only A and B are present, each at 2.00 M . What is the final concentration of A once equi...
Questions

English, 01.07.2019 15:20

English, 01.07.2019 15:20


Biology, 01.07.2019 15:20

Biology, 01.07.2019 15:20

Social Studies, 01.07.2019 15:20

Mathematics, 01.07.2019 15:20




Biology, 01.07.2019 15:20

Mathematics, 01.07.2019 15:20

Health, 01.07.2019 15:20


Health, 01.07.2019 15:20





Mathematics, 01.07.2019 15:20