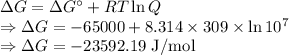Calculate the standard biological Gibbs free energy for the reaction: pyruvate- + NADH + H+(aq) ---> Lactate- + NAD+ at 309 K given that the standard Gibbs free energy = -65.0 kJ/mol at this temperature. This reaction occurs under conditions of low oxygen supply, such as in muscle cells during strenuous exercise. Note: See Box 7.1 on page 164. The biological standard state has hydrogen ions at 1x10-7 molar instead of 1 M.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1
You know the right answer?
Calculate the standard biological Gibbs free energy for the reaction: pyruvate- + NADH + H+(aq) ---&...
Questions






French, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01

History, 05.10.2020 14:01

Chemistry, 05.10.2020 14:01




History, 05.10.2020 14:01



Mathematics, 05.10.2020 14:01



Mathematics, 05.10.2020 14:01

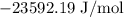
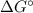 = Standard Gibbs free energy = -65.0 kJ/mol
= Standard Gibbs free energy = -65.0 kJ/mol![[H^+]](/tpl/images/0870/7831/07acb.png) = Biological standard state has hydrogen ions =
= Biological standard state has hydrogen ions = 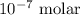
![Q=\dfrac{1}{[H^+]}\\\Rightarrow Q=\dfrac{1}{10^{-7}}\\\Rightarrow Q=10^7](/tpl/images/0870/7831/5fb93.png)