1 point
Why DOESN'T the equation pictured satisfy the Law of Conservation of
Matter?
H2...

Chemistry, 07.11.2020 07:00 edgartorres5123
1 point
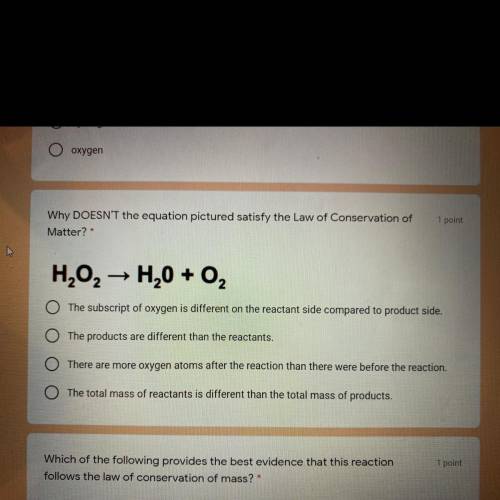
Why DOESN'T the equation pictured satisfy the Law of Conservation of
Matter?
H202 → H20 + O2
The subscript of oxygen is different on the reactant side compared to product side.
The products are different than the reactants.
There are more oxygen atoms after the reaction than there were before the reaction.
The total mass of reactants is different than the total mass of products.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1


Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3
You know the right answer?
Questions


Arts, 05.01.2021 17:40




Chemistry, 05.01.2021 17:40


Computers and Technology, 05.01.2021 17:40

Computers and Technology, 05.01.2021 17:40


English, 05.01.2021 17:40

Mathematics, 05.01.2021 17:40


History, 05.01.2021 17:40

Mathematics, 05.01.2021 17:40

Social Studies, 05.01.2021 17:40

Mathematics, 05.01.2021 17:40


Mathematics, 05.01.2021 17:40

Biology, 05.01.2021 17:40



