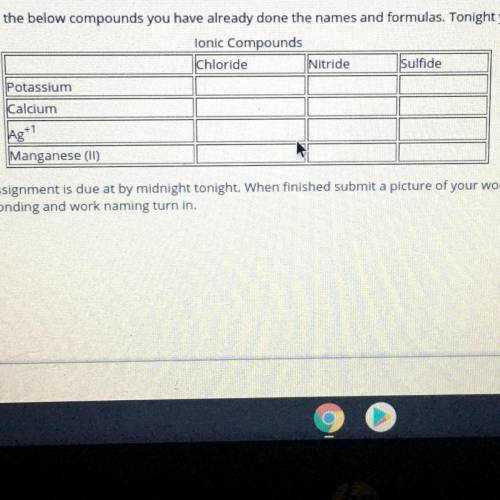
I’ll mark brainliest please
...

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2

You know the right answer?
Questions

History, 06.01.2020 21:31



English, 06.01.2020 21:31

English, 06.01.2020 21:31

Mathematics, 06.01.2020 21:31

Biology, 06.01.2020 21:31


Chemistry, 06.01.2020 21:31




Engineering, 06.01.2020 21:31



Computers and Technology, 06.01.2020 21:31


Business, 06.01.2020 21:31