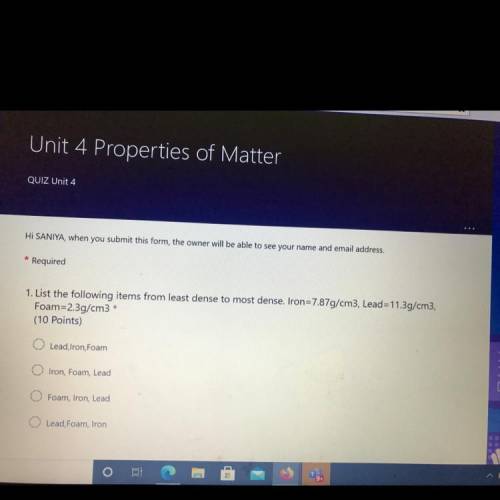
:answer question number 1
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 12:50
Use the standard enthalpies of formation for the reactants and products to solve for the δhrxn for the following reaction. (the δhf of c2h4 is 52.26 kj/mol, co2 is -393.509 kj/mol, and h2o is -241.818 kj.) c2h4 (g) + 3o2(g) 2co2 (g) + 2h2o(g) δhrxn = the reaction is .
Answers: 3

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

You know the right answer?
Questions


Mathematics, 01.04.2021 19:50



Mathematics, 01.04.2021 19:50

History, 01.04.2021 19:50


History, 01.04.2021 19:50


Mathematics, 01.04.2021 19:50

Mathematics, 01.04.2021 19:50

Health, 01.04.2021 19:50

English, 01.04.2021 19:50

Mathematics, 01.04.2021 19:50


Mathematics, 01.04.2021 19:50

Mathematics, 01.04.2021 19:50