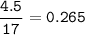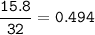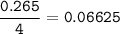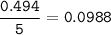Chemistry, 11.11.2020 06:10 sofyan00404
The chemical equation represents a reaction between ammonia and oxygen to form nitrogen monoxide and water. 4 N H 3 ( g ) + 5 O 2 ( g ) → 4 NO ( g ) + 6 H 2 O ( g ) What is the limiting reagent when a 4.50 g sample of ammonia is mixed with 15.80 g of oxygen?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
What was the procedure by which case united states vs lopez went to court
Answers: 1

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1
You know the right answer?
The chemical equation represents a reaction between ammonia and oxygen to form nitrogen monoxide and...
Questions

Mathematics, 05.05.2020 16:26

Mathematics, 05.05.2020 16:26


Physics, 05.05.2020 16:26



Health, 05.05.2020 16:26



Biology, 05.05.2020 16:26


Mathematics, 05.05.2020 16:26

Chemistry, 05.05.2020 16:26




English, 05.05.2020 16:26



Chemistry, 05.05.2020 16:26