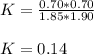Chemistry, 11.11.2020 17:50 karlacarreras152
For the following reaction at equilibrium SO3(g) + NO(g) = SO2(g) + NO2(g)It is found that [SO2] = 0.70 M and [NO] = 1.20 M. Calculate the equilibrium constant for the readction knowing that the initial concentration were [SO3] = 2.55 M and [NO] = 1.90 M.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2
You know the right answer?
For the following reaction at equilibrium SO3(g) + NO(g) = SO2(g) + NO2(g)It is found that [SO2] = 0...
Questions

Mathematics, 24.12.2020 02:00





Chemistry, 24.12.2020 02:00




Mathematics, 24.12.2020 02:00


Mathematics, 24.12.2020 02:10

English, 24.12.2020 02:10



Chemistry, 24.12.2020 02:10


Mathematics, 24.12.2020 02:10


Mathematics, 24.12.2020 02:10

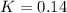
![K=\frac{[SO_2][NO_2]}{[SO_3][NO]}](/tpl/images/0887/9424/17768.png)
 is:
is: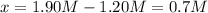
![K=\frac{x*x}{([SO_3]_0-x)([NO]_0-x)}](/tpl/images/0887/9424/71b6f.png)
![[SO_3]=2.55M-0.70M=1.85M](/tpl/images/0887/9424/debed.png)
![[NO]=1.20M](/tpl/images/0887/9424/e6bfc.png)
![[SO_2]=0.70M](/tpl/images/0887/9424/ae391.png)
![[NO_2]=0.70M](/tpl/images/0887/9424/98697.png)