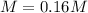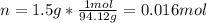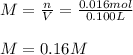Chemistry, 11.11.2020 17:50 jessica6597
Phenol (C6H5OH) is often used as an antiseptic in mouthwashes and throat lozenges. If a mouthwash has a phenol concentration of 1.5 g per 100 mL of solution, what is the molarity of phenol?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 08:30
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1
You know the right answer?
Phenol (C6H5OH) is often used as an antiseptic in mouthwashes and throat lozenges. If a mouthwash ha...
Questions

Biology, 29.10.2019 09:31

Mathematics, 29.10.2019 09:31

Chemistry, 29.10.2019 09:31

Computers and Technology, 29.10.2019 09:31



History, 29.10.2019 09:31

English, 29.10.2019 09:31

Health, 29.10.2019 09:31


History, 29.10.2019 09:31




Mathematics, 29.10.2019 09:31

English, 29.10.2019 09:31

Mathematics, 29.10.2019 09:31

Biology, 29.10.2019 09:31

History, 29.10.2019 09:31