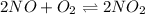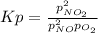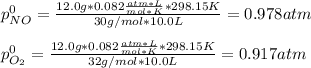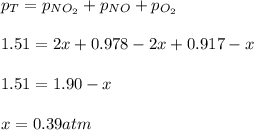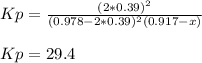Chemistry, 11.11.2020 17:50 dillon3466
For the reaction of nitric oxide and oxygen to form nitrogen dioxide, the reaction begins with 12.0 g of nitric oxide and 12.0 g of oxygen at 25oC in a 10.0 L container. At equilibrium, the pressure in the container is 1148 mmHg, what is Kp?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1


Chemistry, 23.06.2019 04:00
Which of these are physical changes in matter? check all that apply boiling water a pencil being sharpened exploding dynamite freezing water rotting cheese
Answers: 1

Chemistry, 23.06.2019 08:00
Suppose a pair of chemical compounds a and b can react in two different ways: a + b -> c reaction 1 gives product c. a + b -> d reaction 2 gives product d. the following facts are known about the two reactions: . reaction 1 is endothermic and reaction 2 is exothermic. if a reaction vessel is charged (filled) with a and b , then at first d is produced faster than c. use these facts to sketch a qualitative reaction energy diagram for both reactions. note: because these sketches are only qualitative, the energies don? t have to be exact. they only have to have the right relationship to each other. for example, if one energy is less than another, that fact should be clear in your sketch.
Answers: 3
You know the right answer?
For the reaction of nitric oxide and oxygen to form nitrogen dioxide, the reaction begins with 12.0...
Questions

English, 25.04.2021 07:20




Biology, 25.04.2021 07:20

English, 25.04.2021 07:30

Mathematics, 25.04.2021 07:30


Spanish, 25.04.2021 07:30


Biology, 25.04.2021 07:30





Business, 25.04.2021 07:30

Mathematics, 25.04.2021 07:30

History, 25.04.2021 07:30