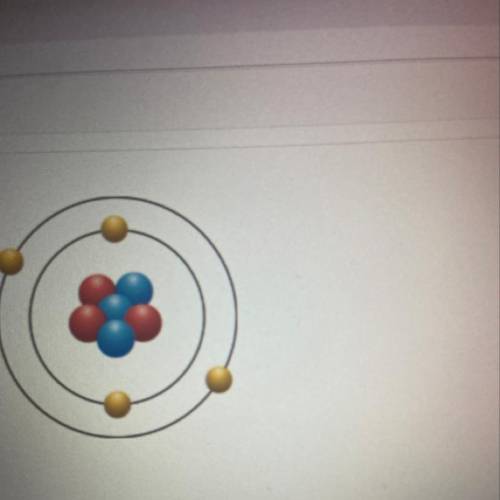
What element is represented in this model of the atomic number is 3
This is science
...


Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Read the given expression. x = number of protons − number of core electrons which of the following explains the identity of x and its trends across a period? x is the effective nuclear charge, and it remains constant across a period. x is the screening constant, and it remains constant across a period. x is the effective nuclear charge, and it increases across a period. x is the screening constant, and it increases across a period.
Answers: 1

Chemistry, 21.06.2019 21:10
Identify one disadvantage to each of the following models of electron configuration: dot structures arrow and line diagrams written electron configurations type in your answer below.
Answers: 1

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

You know the right answer?
Questions


Engineering, 12.01.2021 16:50


Computers and Technology, 12.01.2021 16:50



Mathematics, 12.01.2021 16:50

Mathematics, 12.01.2021 16:50



English, 12.01.2021 16:50



Biology, 12.01.2021 16:50



Biology, 12.01.2021 16:50


English, 12.01.2021 16:50