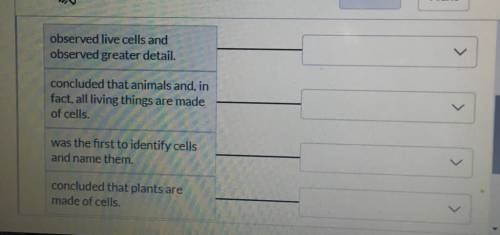
What is the answer? Please
...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1


Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
Questions

Chemistry, 01.09.2019 18:50





History, 01.09.2019 18:50

Social Studies, 01.09.2019 18:50



Computers and Technology, 01.09.2019 18:50

History, 01.09.2019 18:50

Chemistry, 01.09.2019 18:50


History, 01.09.2019 19:00

Mathematics, 01.09.2019 19:00

Mathematics, 01.09.2019 19:00



Physics, 01.09.2019 19:00

English, 01.09.2019 19:00