
Chemistry, 13.11.2020 06:20 msmojangles
Assume a density of water of 1.00 g/mL, and calculate the mass of water in the solution

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2


Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3
You know the right answer?
Assume a density of water of 1.00 g/mL, and calculate the mass of water in the solution...
Questions

History, 18.10.2019 19:00


Chemistry, 18.10.2019 19:00




History, 18.10.2019 19:00

Chemistry, 18.10.2019 19:00

Arts, 18.10.2019 19:00

Mathematics, 18.10.2019 19:00

Mathematics, 18.10.2019 19:00




History, 18.10.2019 19:00



Biology, 18.10.2019 19:00


History, 18.10.2019 19:00

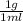 so from this equation, you will get 1 g and you can to SI to be
so from this equation, you will get 1 g and you can to SI to be 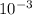 kg
kg

