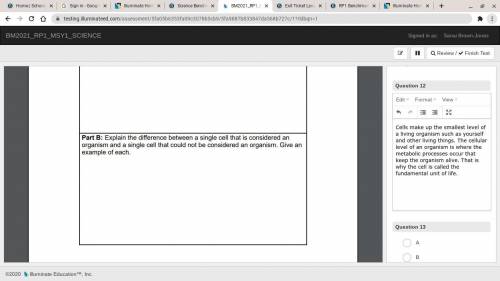
Read the screenshot and HELP
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2


Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3
You know the right answer?
Questions


History, 11.09.2019 01:20


Biology, 11.09.2019 01:20

Mathematics, 11.09.2019 01:20

Mathematics, 11.09.2019 01:20



Mathematics, 11.09.2019 01:20

Health, 11.09.2019 01:20

Social Studies, 11.09.2019 01:20

Health, 11.09.2019 01:20


Physics, 11.09.2019 01:20


Mathematics, 11.09.2019 01:20

History, 11.09.2019 01:20


Geography, 11.09.2019 01:20

Mathematics, 11.09.2019 01:20