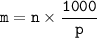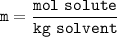Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
mol of water?
A. Convert the 200 mol of water to kilograms of water.
B. Convert the 200 mol of water to liters of water.
C. Convert the 10 mol of NaCl to grams of NaCl.
O D. Convert the 10 mol of NaCl to kilograms of NaCl.
SUBMIT

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:00
How are ionic bonds formed and what is the attractive force within an ionic bond
Answers: 1

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2
You know the right answer?
Question 1 of 10
What would you need to do to calculate the molality of 10 mol of NaCl in 200
...
...
Questions


History, 14.04.2020 00:34



Mathematics, 14.04.2020 00:34

Mathematics, 14.04.2020 00:34

Biology, 14.04.2020 00:34



Mathematics, 14.04.2020 00:34

Mathematics, 14.04.2020 00:34


Chemistry, 14.04.2020 00:34



Mathematics, 14.04.2020 00:34



Mathematics, 14.04.2020 00:34