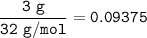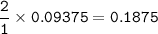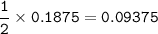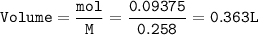Chemistry, 15.11.2020 23:10 scadengo123
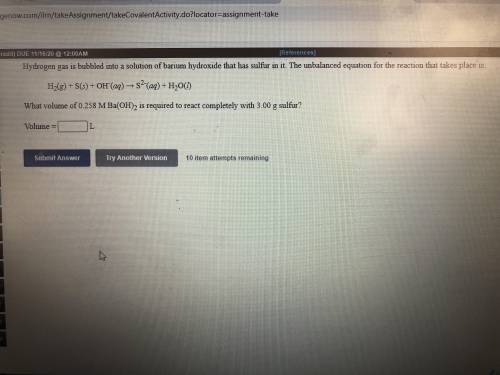
Hydrogen gas is bubbled into a solution of barium hydroxide that has sulfur in it. The unbalanced equation for the reaction that takes place is: H2+S+OH- —-> S2- + H2O What volume of 0.258 M Ba(OH)2 is required to react completely with 3.00g sulfur?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
Hydrogen gas is bubbled into a solution of barium hydroxide that has sulfur in it. The unbalanced eq...
Questions


Mathematics, 28.11.2019 15:31

English, 28.11.2019 15:31

History, 28.11.2019 15:31

Health, 28.11.2019 15:31



English, 28.11.2019 15:31

Mathematics, 28.11.2019 15:31

Health, 28.11.2019 15:31



Biology, 28.11.2019 15:31





Mathematics, 28.11.2019 15:31