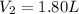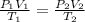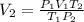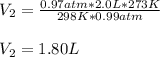A chemist conducts an experiment in which 2.0 L of hydrogen gas is collected over water at 1 atm and 298 K. Remember the pressure due to the hydrogen gas is 0.97 atm. Using the combined gas law, calculate the volume in L of the dry hydrogen gas at 273 K and a pressure of 1 atm.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1


Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3
You know the right answer?
A chemist conducts an experiment in which 2.0 L of hydrogen gas is collected over water at 1 atm and...
Questions

Mathematics, 28.06.2019 18:40

Physics, 28.06.2019 18:40




Mathematics, 28.06.2019 18:40



History, 28.06.2019 18:40

Biology, 28.06.2019 18:40

English, 28.06.2019 18:40

Mathematics, 28.06.2019 18:40





Mathematics, 28.06.2019 18:40


English, 28.06.2019 18:40

Physics, 28.06.2019 18:40