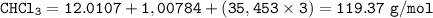What is the molar mass of CHCl3?
48.47 g/mol
83.92 g/mol
119.37 g/mol
121.39...

Chemistry, 16.11.2020 19:20 tashanicole
What is the molar mass of CHCl3?
48.47 g/mol
83.92 g/mol
119.37 g/mol
121.39 g/mol

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Questions



English, 06.04.2020 16:27






Mathematics, 06.04.2020 16:27

Mathematics, 06.04.2020 16:27

History, 06.04.2020 16:27


Mathematics, 06.04.2020 16:27


Chemistry, 06.04.2020 16:27




Mathematics, 06.04.2020 16:27