
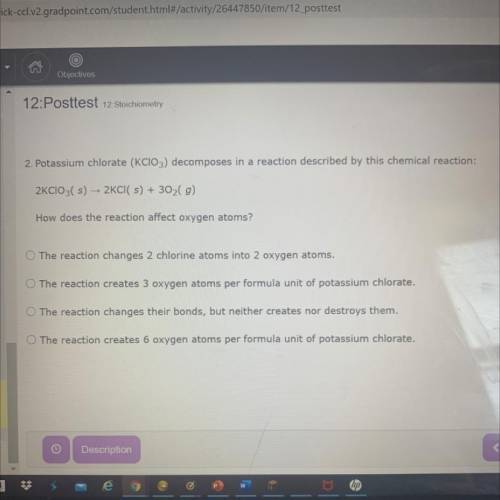
Potassium chlorate (KClO3) decomposes in a reaction described by this chemical reaction:
2KCIO3( s) - 2KCI( s) + 302(g)
How does the reaction affect oxygen atoms?
A. The reaction changes 2 chlorine atoms into 2 oxygen atoms.
B. The reaction creates 3 oxygen atoms per formula unit of potassium chlorate.
C. The reaction changes their bonds, but neither creates nor destroys them.
D. The reaction creates 6 oxygen atoms per formula unit of potassium chlorate.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Balance this equation: n2 + h2 > nh3, write the following molar ratios: n2 / n2 / nh3 h2 /
Answers: 1

Chemistry, 21.06.2019 18:30
Which orbitals form a pi bond? a.the s orbital and three p orbitals b.the s orbital and two p orbitals c.overlapping p orbitals d.overlapping hybrid orbitals
Answers: 2


Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1
You know the right answer?
Potassium chlorate (KClO3) decomposes in a reaction described by this chemical reaction:
2KCIO3( s)...
Questions

English, 03.03.2021 17:20





English, 03.03.2021 17:20

Mathematics, 03.03.2021 17:20


English, 03.03.2021 17:20


English, 03.03.2021 17:20

Mathematics, 03.03.2021 17:20



Mathematics, 03.03.2021 17:20

Social Studies, 03.03.2021 17:20



Mathematics, 03.03.2021 17:20



