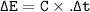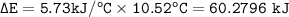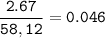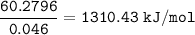Chemistry, 17.11.2020 05:40 jnannsbwnw4216
2.67 grams of butane (C4H10) is combusted in a bomb calorimeter. The temperature increases from 25.68 C to 36.2C. What is the change in the internal energy (deltaE) in KJ/mol for the reaction if the heat capacity of the bomb calorimeter is 5.73 kJ/C?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2


You know the right answer?
2.67 grams of butane (C4H10) is combusted in a bomb calorimeter. The temperature increases from 25.6...
Questions

Mathematics, 18.05.2020 08:57



Mathematics, 18.05.2020 08:57



Mathematics, 18.05.2020 08:57

History, 18.05.2020 08:57



Mathematics, 18.05.2020 08:57

Biology, 18.05.2020 08:57



Mathematics, 18.05.2020 08:57



History, 18.05.2020 08:57