
Copper metal (Cu) reacts with silver nitrate (AgNO3) in aqueous solution to form Ag and Cu(NO3)2. An excess of AgNO3 is present. The balanced chemical equation is shown below.
Cu + 2AgNO3 Right arrow. Cu(NO3)2 + 2Ag
The molar mass of Cu is 63.5 g/mol. The molar mass of Ag is 107.9 g/mol. What mass, in grams, of Ag is produced from reaction of 31.75 g of Cu?
26.95
107.9
215.91
431.82

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2
You know the right answer?
Copper metal (Cu) reacts with silver nitrate (AgNO3) in aqueous solution to form Ag and Cu(NO3)2. An...
Questions

Social Studies, 30.07.2019 14:00

Biology, 30.07.2019 14:00

Social Studies, 30.07.2019 14:00


Social Studies, 30.07.2019 14:00



Chemistry, 30.07.2019 14:00


Social Studies, 30.07.2019 14:00


Social Studies, 30.07.2019 14:00


Business, 30.07.2019 14:00



Social Studies, 30.07.2019 14:00


Social Studies, 30.07.2019 14:00

Social Studies, 30.07.2019 14:00

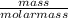
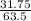 = 0.5moles
= 0.5moles 

