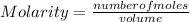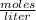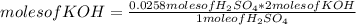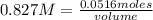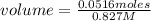Chemistry, 18.11.2020 17:10 ahmedeldyame
In a titration of 35.00 mL of 0.737 M H2SO4, mL of a 0.827 M KOH solution is required for neutralization.
A) 35.0
B) 1.12
C) 25.8
D) 62.4
E) 39.3

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 10:40
Asolid that forms and separates from a liquid mixture is called
Answers: 2

You know the right answer?
In a titration of 35.00 mL of 0.737 M H2SO4, mL of a 0.827 M KOH solution is required for neutraliz...
Questions












Mathematics, 20.02.2020 22:49

Social Studies, 20.02.2020 22:49







English, 20.02.2020 22:50