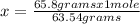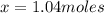B 0 10.9 mol Cu

Chemistry, 18.11.2020 23:50 deedee2616
How many moles of copper (Cu) are in 65.8 g Cu? (3 points)
A 01.04 mol Cu
B 0 10.9 mol Cu
C 41.7 mol Cu
D 63.5 mol Cu

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1


Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2
You know the right answer?
How many moles of copper (Cu) are in 65.8 g Cu? (3 points)
A 01.04 mol Cu
B 0 10.9 mol Cu
B 0 10.9 mol Cu
Questions




Mathematics, 30.09.2019 17:10


Mathematics, 30.09.2019 17:10

Health, 30.09.2019 17:10

Mathematics, 30.09.2019 17:10



English, 30.09.2019 17:10




Mathematics, 30.09.2019 17:10





English, 30.09.2019 17:10

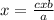
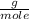 , this is the amount of mass a substance contains in one mole.
, this is the amount of mass a substance contains in one mole.