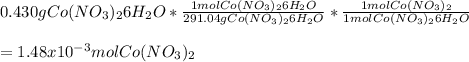Chemistry, 20.11.2020 14:00 meramera50
If you mass 0.430 g of Co(NO3)2 6H2O and dissolve it in water, what further information would you need to calculate:.
a. the number of moles of Co(NO3)2 in the solution?
b. the molarity of the solution

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2
You know the right answer?
If you mass 0.430 g of Co(NO3)2 6H2O and dissolve it in water, what further information would you ne...
Questions



Mathematics, 16.10.2020 15:01


Mathematics, 16.10.2020 15:01

History, 16.10.2020 15:01

History, 16.10.2020 15:01






Social Studies, 16.10.2020 15:01

Chemistry, 16.10.2020 15:01

Health, 16.10.2020 15:01



Mathematics, 16.10.2020 15:01


Mathematics, 16.10.2020 15:01