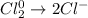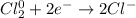Chemistry, 24.11.2020 21:20 torieegrey
The balanced half-reaction in which chlorine gas is reduced to the aqueous chloride ion is a process. one-electron two-electron four-electron three-electron six-electron

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
The balanced half-reaction in which chlorine gas is reduced to the aqueous chloride ion is a proces...
Questions

Mathematics, 01.09.2021 04:30

English, 01.09.2021 04:30

Mathematics, 01.09.2021 04:30


Mathematics, 01.09.2021 04:30

Mathematics, 01.09.2021 04:30






Social Studies, 01.09.2021 04:30

History, 01.09.2021 04:30

Chemistry, 01.09.2021 04:30


English, 01.09.2021 04:30


Mathematics, 01.09.2021 04:30

Mathematics, 01.09.2021 04:30

Mathematics, 01.09.2021 04:30