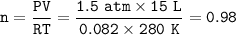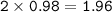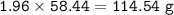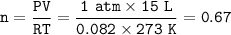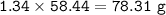Part 1. A chemist reacted 15.0 liters of F2 gas with NaCl in the laboratory to form Cl2 and NaF. Use the ideal gas law equation to determine the
mass of NaCl that reacted with F2 at 280. K and 1.50 atm.
F2 + 2NaCl + Cl2 + 2NaF
Part 2. Explain how you would determine the mass of sodium chloride that can react with the same volume of fluorine gas at STP

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which is true of the reactants in this displacement reaction? fe + 2hcl fecl2 + h2 a. the reactants are located to the left of the arrow in the chemical equation. b. the reactants contain 1 iron atom, 2 hydrogen atoms, and 1 chlorine atom. c. the reactants are the atoms, molecules, or compounds formed in the reaction. d. the reactants have the same physical and chemical properties as the products.
Answers: 1

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1

Chemistry, 22.06.2019 17:00
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3
You know the right answer?
Part 1. A chemist reacted 15.0 liters of F2 gas with NaCl in the laboratory to form Cl2 and NaF. Use...
Questions


English, 12.10.2019 02:00



Mathematics, 12.10.2019 02:00

Mathematics, 12.10.2019 02:00

Mathematics, 12.10.2019 02:00


Mathematics, 12.10.2019 02:00

Spanish, 12.10.2019 02:00


Mathematics, 12.10.2019 02:00

Mathematics, 12.10.2019 02:00

Mathematics, 12.10.2019 02:00


Mathematics, 12.10.2019 02:00

English, 12.10.2019 02:00

Mathematics, 12.10.2019 02:00


Mathematics, 12.10.2019 02:00