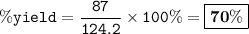Use the equation below to solve the problem that follows.
2H2 (g) + O2 (g) → 2H2O (g)
W...

Chemistry, 26.11.2020 01:00 pinkpearl20
Use the equation below to solve the problem that follows.
2H2 (g) + O2 (g) → 2H2O (g)
When David reacts 13.8 grams of hydrogen gas with excess oxygen, 87.0 grams of water are formed. Calculate his percent yield of water.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
If you want to create an electrical current, which situation would produce a solution capable of this
Answers: 3

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3
You know the right answer?
Questions

Mathematics, 09.12.2019 14:31

Health, 09.12.2019 14:31

Mathematics, 09.12.2019 14:31



Mathematics, 09.12.2019 14:31


Mathematics, 09.12.2019 14:31


History, 09.12.2019 14:31


Social Studies, 09.12.2019 14:31

History, 09.12.2019 14:31




History, 09.12.2019 15:31


Mathematics, 09.12.2019 15:31