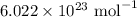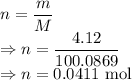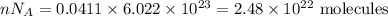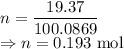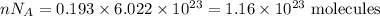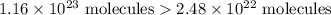I have 2 samples of solid chalk (aka calcium carbonate). Sample A has a total mass of 4.12 g and Sample B has a total mass of 19.37 g. What is the difference between the samples?
A) Sample B has more calcium carbonate molecules
B) Sample B has a larger ratio of carbon, oxygen, and calcium atoms
C) Sample B has more calcium ion than carbonate ions
D) Sample B must have some impurity

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1


Chemistry, 22.06.2019 23:00
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1
You know the right answer?
I have 2 samples of solid chalk (aka calcium carbonate). Sample A has a total mass of 4.12 g and Sam...
Questions



Chemistry, 26.05.2021 06:30

Biology, 26.05.2021 06:30

Social Studies, 26.05.2021 06:30

History, 26.05.2021 06:30


Mathematics, 26.05.2021 06:30


Mathematics, 26.05.2021 06:30





Social Studies, 26.05.2021 06:30



History, 26.05.2021 06:30


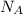 = Avogadro's number =
= Avogadro's number =