Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1


Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
23.1 g of HCl (a strong acid) is added to water to make 1250 mL of solution. Calculate [H3O+] and pH...
Questions


Mathematics, 19.09.2019 00:20

Business, 19.09.2019 00:20


Mathematics, 19.09.2019 00:20

Mathematics, 19.09.2019 00:20




Social Studies, 19.09.2019 00:20


Business, 19.09.2019 00:20


Physics, 19.09.2019 00:20

Mathematics, 19.09.2019 00:20





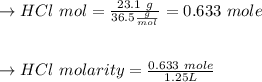
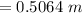
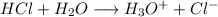
![[H_3O^{+}] =[HCl] \\\\\to [H_3O^{+}] =0.5064 \ m \\\\\to PH =-\log [H_3O^{+}] \\\\](/tpl/images/0929/8717/b345c.png)