
Chemistry, 26.11.2020 07:40 emiller6462
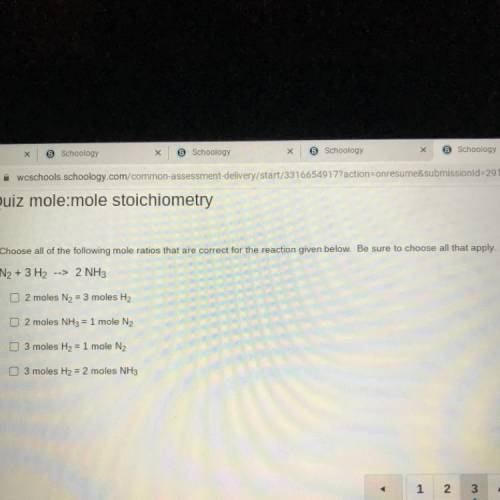
Choose all of the following mole ratios that are correct for the reaction given below. Be sure to choose all that apply.
N2 + 3 H2 --> 2 NH3
2 moles N2 = 3 moles H2
2 moles NH3 = 1 mole N2
3 moles H2 = 1 mole N2
3 moles H2 = 2 moles NH3

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:40
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1


Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
Choose all of the following mole ratios that are correct for the reaction given below. Be sure to ch...
Questions

Chemistry, 14.10.2019 05:50



Mathematics, 14.10.2019 05:50




Geography, 14.10.2019 06:00

Mathematics, 14.10.2019 06:00

History, 14.10.2019 06:00

English, 14.10.2019 06:00



English, 14.10.2019 06:00

Mathematics, 14.10.2019 06:00

Mathematics, 14.10.2019 06:00

History, 14.10.2019 06:00


Mathematics, 14.10.2019 06:00




