Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 23:30
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3

Chemistry, 23.06.2019 03:40
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2

Chemistry, 23.06.2019 07:00
In order for a high temperature boiler or steam engine to produce superheated water, or steam: the heat source must be greater than 100°c the water must be permitted to evaporate quickly the system must be sealed and become pressurized above atmospheric pressure the vapor pressure must be kept below 760 mm(hg)
Answers: 1

Chemistry, 23.06.2019 09:00
Describe the process that was used in this lab to create magnesium oxide, specifically identifying the type of chemical reaction. explain why the product had a higher mass than the reactant, and how this relates to conservation of matter.
Answers: 2
You know the right answer?
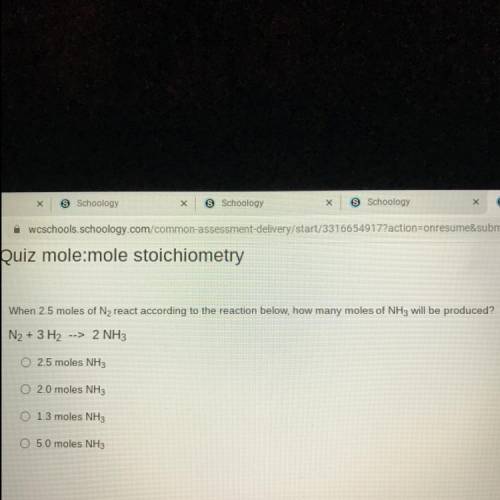
When 2.5 moles of N2 react according to the reaction below, how many moles of NH3 will be produced?...
Questions

History, 26.09.2019 09:30

Mathematics, 26.09.2019 09:30




Mathematics, 26.09.2019 09:30

Biology, 26.09.2019 09:30



History, 26.09.2019 09:30

History, 26.09.2019 09:30

Mathematics, 26.09.2019 09:30

Mathematics, 26.09.2019 09:30

History, 26.09.2019 09:30

History, 26.09.2019 09:30



History, 26.09.2019 09:30