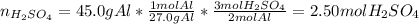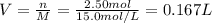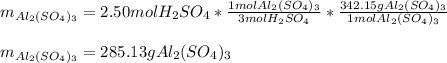Chemistry, 26.11.2020 21:50 JesusisLord2881
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)a. Determine the volume (mL) of 15.0 M sulfuric acid needed to react with 45.0 g of aluminum to produce aluminum sulfate. b. Determine the % yield if 112 g of aluminum sulfate is produced under the above conditions.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3
You know the right answer?
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)a. Determine the volume (mL) of 15.0 M sulfuric acid nee...
Questions



Mathematics, 09.10.2021 01:00




Mathematics, 09.10.2021 01:00


Mathematics, 09.10.2021 01:00

English, 09.10.2021 01:00

Business, 09.10.2021 01:00




Social Studies, 09.10.2021 01:00


Mathematics, 09.10.2021 01:00

Chemistry, 09.10.2021 01:00


Social Studies, 09.10.2021 01:00