DENSITY_9

Chemistry, 26.11.2020 22:30 quigley523
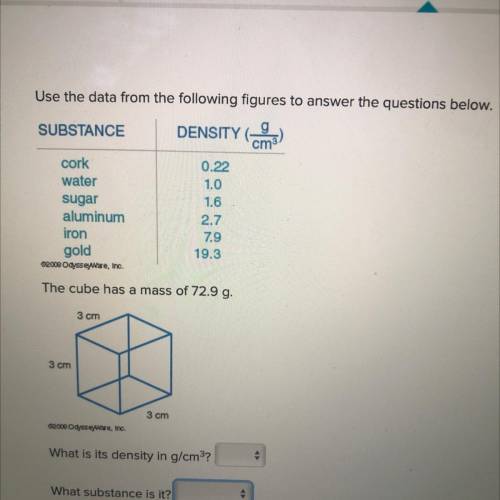
Use the data from the following figures to answer the questions below.
SUBSTANCE
DENSITY_9
cm
cork
0.22
water
1.0
sugar
1.6
aluminum
2.7
79
gold
19.3
02000 Ossewwe, in
The cube has a mass of 72.9 g.
iron
3 cm
3 cm
3 cm
2008 Ogsewu, in
What is its density in g/cm?
What substance is it?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
The density of glycerin is 1.26grams/centimeter cubed . how many is this? use the conversion rates of and . express your answer to the correct number of significant figures.
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1
You know the right answer?
Use the data from the following figures to answer the questions below.
SUBSTANCE
DENSITY_9
DENSITY_9
Questions


Mathematics, 28.10.2019 05:31

History, 28.10.2019 05:31

Mathematics, 28.10.2019 05:31


Health, 28.10.2019 05:31

History, 28.10.2019 05:31




Social Studies, 28.10.2019 05:31




Mathematics, 28.10.2019 05:31



Health, 28.10.2019 05:31

Mathematics, 28.10.2019 05:31

History, 28.10.2019 05:31

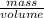
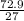 = 2.7g/cm³
= 2.7g/cm³

