
Chemistry, 28.11.2020 07:30 xmanavongrove55
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I have to do is solve through those 2 problems but I'm not sure if I solve the fractions first then multiply that result by 100 or what. I asked my teacher which she helped a little but I don't think she understood where I was lost. If you can help I would really appreciate it. Thank you.
This was the set up question:
4. If 100.0g of nitrogen gas (N2) is reacted with 100.0g of hydrogen gas (H2) to form NH3. What is the limiting and excess reactants?
Hint: Convert grams to moles for each reactant and then convert to moles of NH3. You need your balanced equation from answer 1 to determine the mole relationship between each reactant and the product NH3. Use the periodic table to determine the molar mass of all chemical formulas. Fill in the “?” blanks below to show your work.
and in the screenshot it has everything I'm working with and the conclusions I need to draw from it, I can draw the conclusions just fine on my own but I need help solving.


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I...
Questions




English, 26.02.2020 00:32


History, 26.02.2020 00:32

English, 26.02.2020 00:32

Geography, 26.02.2020 00:32



Mathematics, 26.02.2020 00:32









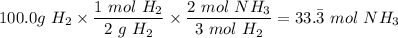
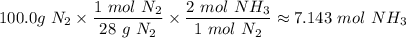
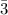 moles of NH₃)
moles of NH₃)

