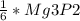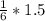Chemistry, 30.11.2020 18:20 descampbell2001
2. 1.5 moles of AgNO3 reacts with 0.5 mole of Mg3P2. Calculate the moles of excess reactant that remains at the end of the reaction. Include math to justify your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Melissa is interested in her family tree and how her family has changed over its many generations. melissa probably more closely resembles
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3
You know the right answer?
2. 1.5 moles of AgNO3 reacts with 0.5 mole of Mg3P2. Calculate the moles of excess
reactant that re...
Questions

Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30


Biology, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30



Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

English, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30



English, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

Mathematics, 09.02.2021 23:30

Chemistry, 09.02.2021 23:30