
Chemistry, 01.12.2020 02:40 santiagobermeo32
4. Chemical formula, mg (Cl0₃)2
a) calculate the number of moles for 3.24g, Mg (Cl03)2
b) calculate the molarity of mg (Cl03)2 if all of it were dissolved in
5.08 L distilled water.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 04:30
This question is about electrolysis. metal spoons can be coated with silver. this is called electroplating. suggest one reason why spoons are electroplated?
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

You know the right answer?
4. Chemical formula, mg (Cl0₃)2
a) calculate the number of moles for 3.24g, Mg (Cl03)2
b) calc...
b) calc...
Questions

English, 20.10.2021 01:00

English, 20.10.2021 01:00

Geography, 20.10.2021 01:00

Mathematics, 20.10.2021 01:00




Mathematics, 20.10.2021 01:00

History, 20.10.2021 01:00

Mathematics, 20.10.2021 01:00

Biology, 20.10.2021 01:00


Mathematics, 20.10.2021 01:00







Mathematics, 20.10.2021 01:00

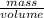
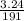 = 0.017mole
= 0.017mole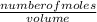
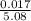 = 0.0033M
= 0.0033M

